Titan® Penile Implants
Performance from the operating room
to the bedroom

Find the right implant for more patients
The prevalence of erectile dysfunction is increasing among men meaning more patients may be seeking a long-term solution.
Inflatable penile prosthesis (IPP) surgery is a big step, and both you and your patient want to make sure it goes smoothly and meets expectations for a satisfying outcome. Complex patient populations can result in a higher likelihood of complications which means as a physician you have a lot to consider when choosing the right implant for your patient.
Operating room considerations
![]()
Up to
78%
of IPP patients are diabetic, which can almost double the risk of infection.1,2
![]()
Up to
50%
of IPP patients had previous pelvic surgery/trauma which may lead to a higher risk of penile shortening and could require special considerations for reservoir placement.4,5
![]()
Up to
33%
of IPP patients had Peyronie’s disease, increasing risk of device malfunction and penile shortening—reducing functionality and post-op satisfaction rates by 7%.1,3,5
![]()
Approximately
30% revision surgeries
Approximately 30% of IPPs implanted in 2021 were revision surgeries, also increasing likelihood of infection among other possible complications.5,6
*Note that many etiologies are concomitant and will not equal 100%
Bedroom considerations
Along with complexities in the operating room there are patient concerns that carry into the bedroom.
![]()
The number one concern among patients is decrease in penile size
70%
of of patients endorse loss of length even in absence of measurable evidence.5,8
![]()
Patient dissatisfaction after surgery accounts for approximately
75%
of malpractice claims against surgeons, with average payouts in urology of around $319,000.5,7
![]()
Titan® IPP satisfaction rate
96%+
IPPs have a high satisfaction rate for both patients and their partners.9
![]()
Coloplast Titan® IPP:
Built to perform.
Designed to satisfy.
Titan IPP provides performance in both the operating room and the bedroom. Titan IPP is designed to overcome complications, address patient satisfaction, and allow you to serve the diverse needs of more patients that walk through your door.
Titan IPP performance in the operating room
Technology that’s designed and engineered for performance

Power of choice
HydroVANTAGE™ hydrophilic coating absorbs an aqueous solution of physician’s choice on submerging the device in the solution.
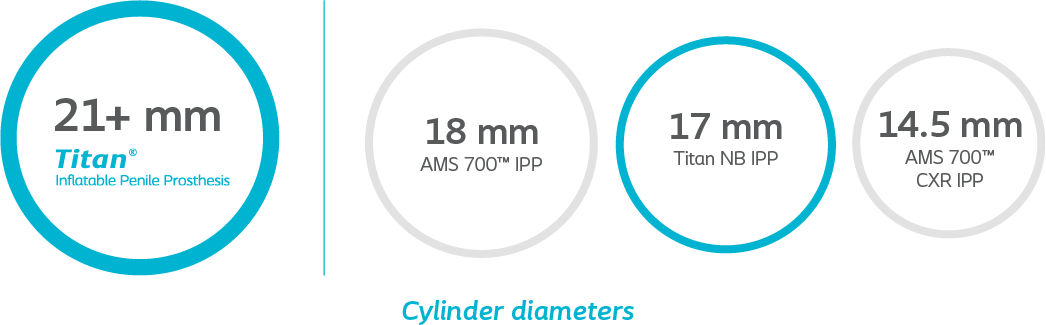
Tailored fit
Because there’s a wide variety in the male anatomy, Titan IPP is available in a wide range of standard and narrow sizes so you can choose the right fit for each patient.
Cylinder diameters are not shown to scale.
*AMS 700™ is a trademark of Boston Scientific Corporation.

Titan IPP performance in the bedroom

Performance that satisfies so your patients can rediscover their confidence and get back to a healthy sex life
17%
MORE GIRTH10
compared to AMS 700™ CX IPP
Maximizing girth may lead to increased partner stimulation and satisfaction.
3x
MORE RIGID11
compared to AMS 700™ LGX IPP
Increased rigidity reduces the chance of buckling during sex.
3x
MORE DURABLE12
compared to AMS 700™ CX IPP
Enhanced penetration performance for more confidence in a variety of sexual positions so patients can feel uninhibited.
*AMS 700™ is a trademark of Boston Scientific Corporation

Two pump designs to better suit patient needs
The tried-and-true Titan pump has decades of experience, and the Titan Touch Pump offers simplified deflation with less force required to trigger device inflation.

Soft-molded tips
For a more natural feel that more closely resemble human corporal tip.
Lock-out valve
keeps the patient in control by preventing the embarrassment of unintended auto-inflation.
-
Clinician Videos
Titan Touch IPP Animation Share
Patient Inflate/Deflate Demonstration Video Share
Clinical Evidence
Inflatable Penile Prosthesis Placement in Men with Peyronie’s Disease and Drug-resistance Erectile Dysfunction: A Single-center Study Share
Optimal Modeling: an Updated Method for Safely and Effectively Eliminating Curvature During Penile Prosthesis Implantation Share
Long-term Follow-up of Treatment for Peyronie’s Disease: Modeling the Penis Over an Inflatible Penile Prosthesis Share
Other Resources
Titan Penile Implant Dictation Form Share
MRI Compatibility and Latex Content Memo (Penile Prostheses) Share
2024 Men’s Health US Coding and Payment Reference Guide – Physician Share
2024 Men’s Health US Coding and Payment Reference Guide – Hospital Outpatient and Ambulatory Surgery Center Share
Clinician Resources
Clinician Videos
Titan IPP Device Prep Send a SMS Share
Titan IPP Animation Send a SMS Share
Titan Touch IPP Animation Send a SMS Share
Patient Inflate/Deflate Demonstration Video Send a SMS Share
Penoscrotal Placement of Titan IPP Send a SMS Share
Infrapubic Placement of Titan IPP Send a SMS Share
Clinical Evidence
Inflatable Penile Prosthesis Placement in Men with Peyronie’s Disease and Drug-resistance Erectile Dysfunction: A Single-center Study Send a SMS Share
Optimal Modeling: an Updated Method for Safely and Effectively Eliminating Curvature During Penile Prosthesis Implantation Send a SMS Share
Long-term Follow-up of Treatment for Peyronie’s Disease: Modeling the Penis Over an Inflatible Penile Prosthesis Send a SMS Share
Patient and Partner Satisfaction after Penile Implant Send a SMS Share
Other Resources
Titan Penile Implant Dictation Form Send a SMS Share
MRI Compatibility and Latex Content Memo (Penile Prostheses) Send a SMS Share
2024 Men’s Health US Coding and Payment Reference Guide – Physician Send a SMS Share
2024 Men’s Health US Coding and Payment Reference Guide – Hospital Outpatient and Ambulatory Surgery Center Send a SMS Share
Patient Resources
Ordering information
For ordering information, please call 800-258-3476.
