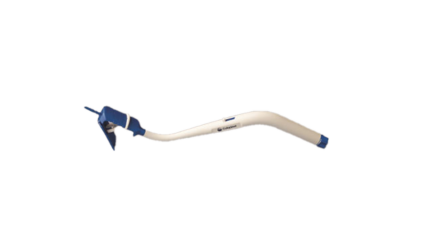
Meridian® Vaginal Positioning System
The Meridian Vaginal Positioning System is intended for use in general gynecological surgery to assist in the position and manipulation of the vagina. The device can be used with tactile feedback and/or direct visualization during surgical procedures such as sacrocolpopexy.
Ordering Information
For ordering information, please call 800.258.3476.
Meridian® VPS™ Brief Statement
Indications
The Meridian VPS is a single use device intended to assist in the position and manipulation of the vagina during gynecologic surgical procedures such as sacrocolpopexy.
Contraindications
The Meridian VPS is contraindicated for use in patients with the following conditions:
- Pregnancy
- Intrauterine Device (IUD) present
- Physician deems use inadvisable
Warnings
The procedure to insert the device requires a good knowledge of local anatomy and the correct use of the manipulator in order to avoid perioperative damage to adjacent anatomical structures.
Avoid using excessive force during vaginal insertion, manipulation and removal of the device.
Precautions
Reuse of this single use product may create a potential harm to the physician, medical staff and/or patient.
To reduce the risk of perforation, if any resistance is felt when inserting the vaginal positioning system, do not force the vaginal positioning system against the resistance.
Potential Complications
Potential complications associated with vaginal manipulators include adverse tissue reaction, bleeding, cramping or discomfort, damage to blood vessels, nerves, connective tissue and other adjacent structures, infection, muscle spasms, organ perforation and injury, pain, and tissue damage.
The information provided is not comprehensive with regard to product risks. For a comprehensive listing of indications, contraindications, warnings, precautions, and adverse events refer to the product’s Instructions for Use. Alternatively, you may contact a Coloplast representative at 1-800-258-3476 and/or visit the company Website at www.coloplast.com.
Caution: Federal law (USA) restricts this device to sale by or on the order of a physician.
Minneapolis, MN
06/01/2018
PM-04816
Ordering information
For ordering information, please call 800-258-3476.