Dormia® Front Nitinol Hybrid Grasper
Dormia® Front nitinol hybrid graspers are designed for use during flexible ureteroscopy within the kidney and ureter. The snare-like design permits capture, repositioning, and removal of stones as small as 1mm in a single procedure. 1.5 Fr diameter baskets are an effective option for use in the lower pole of the kidney because of their ability to increase irrigation, improve visualization, and extend scope deflection compared to larger diameter baskets.
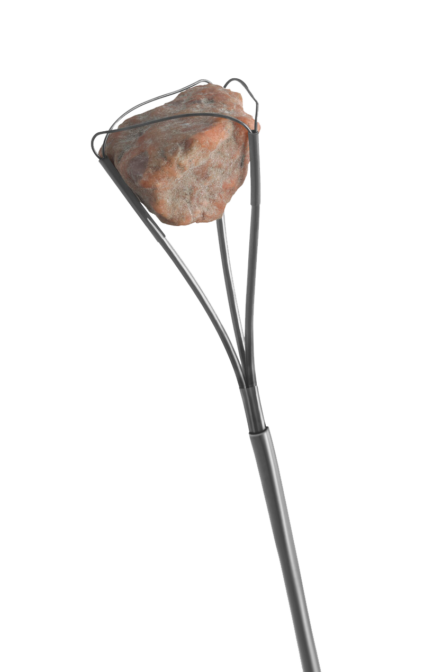
Quick Look
Multifunctional Basket
- Snare-like design permits capture, repositioning, and removal of stones as small as 1 mm
- Reinforced tip with shrink tubing made of polyester for durability
- Nitinol material has excellent kink-resistance and shape memory characteristics
Smooth Gliding Sheath with Small Diameter Option
- Polyamide sheath to facilitate glide and smoother movement. Two diameters: 1.5 and 2Ch/Fr
- 1.5 Fr diameter baskets are an effective option for use in the lower pole of the kidney because of their ability to increase irrigation, improve visualization, and extend scope deflection compared to larger diameter baskets
- 1.5 Fr Dormia® Front permits simultaneous placement of a 272 micron laser fiber within the working channel of the scope
Durable Construction Handle
- Allows the basket to be opened and closed multiple times during a procedure
- Engraved with open and close directions
Ordering Information
| Item | Description | Diameter | Basket Size | Length | Sales UOM | EA/Sales UOM |
| DOF158 | Dormia® Front Nitinol Hybrid Basket 1.5 Fr 8 mm | 1.5 Fr | 8 mm | 120 cm | EA | 1 |
| DOF151 | Dormia® Front Nitinol Hybrid Basket 1.5 Fr 11 mmm | 1.5 Fr | 11 mm | 120 cm | EA | 1 |
| DOF208 | Dormia® Front Nitinol Hybrid Basket 2 Fr 8 mm | 2 Fr | 8 mm | 120 cm | EA | 1 |
| DOF201 | Dormia® Front Nitinol Hybrid Basket 2 Fr 11 mm | 2 Fr | 11 mm | 120 cm | EA | 1 |
Dormia® Front Nitinol Hybrid Basket Brief Statement
Indications
For stone removal during the course of rigid and flexible ureterorenoscopy, cystoscopy, endoscopic retrograde cholangioscopy (ERC), endoscopic retro- grade cholangiopancreatography (ERCP).
Intended Purpose
Stone retrieval devices serve for the endoscopic removal of stones and their fragments from the urogenital and gastroenterological tract during the course of retrograde interventions.
Contraindications
The contraindications of the above endoscopic interventions apply. The stone retrieval devices may not be used for Percutaneous Nephrolithotomy (PCNL). Stone retrieval devices may not be used for intravascular applications or other application areas, as sufficient clinical experience is lacking for this.
Warnings and Precautions
If used improperly, stone retrieval devices can cause the perforation of tissue, in particular if the stones are lodged on the vessel wall. The stone bed is then frequently very fragile. The use of contrast media can lead to adhesions that can limit the functionality of the stone retrieval device. Some stones may be too large to be removed with the stone retrieval device through the endoscope because the stone could get stuck in the working channel of the endoscope during removal. Therefore, always the complete system of endoscope and retrieval device shall be removed and the retrieval device shall be emptied outside the human body. Stone retrieval devices may not be used for mechanical stone crushing (lithotripsy). This type of device must be used only by trained and experienced professionals. Do not use the stone retrieval device if the stone is too large. If excessive force is used, there is a potential for vessel wall tear.
Potential Complications
The following complications are possible when using stone retrieval devices for stone removal:
- Entrapment of large stones
- Inability to disengage the dislodger from irretrievable stones requiring the application of other interventions
- Tissue perforation
- Breakage of the stone retrieval device
- Infection
- Non-retrievable stones
The risks and benefits of using Dormia® Front Stone Extractor should be considered in patients.
The information provided is not comprehensive with regard to product risks. For a comprehensive listing of indications, contraindications, warnings and precautions refer to the product’s Instructions for Use. Alternatively, you may contact a Coloplast representative at 1-800-258-3476 and/or visit the company Website at www.coloplast.com.
Complications from the use of this device should be brought to the attention of your Coloplast Representative and your physician.
Caution: Federal (USA) law restricts this device to sale by or on the order of a physician.
-
Clinical Summaries
Impact on Active Scope Deflection and Irrigation Flow of All Endoscopic Working Tools during Flexible Ureteroscopy Clinical Summary Share
Size does matter: 1.5 Fr. stone baskets almost double irrigation flow during flexible ureteroscopy compared to 1.9 Fr. Stone baskets Clinical Summary Share
Resources
Marketing Materials
Dormia® Front Information Sheet Send a SMS Share
Dormia® Product Brochure Send a SMS Share
Videos
Dormia® Front Animation (English) – PM-17212 Send a SMS Share
White Papers
Dormia® Bench Test White Paper Send a SMS Share
Clinical Summaries
Impact on Active Scope Deflection and Irrigation Flow of All Endoscopic Working Tools during Flexible Ureteroscopy Clinical Summary Send a SMS Share
Size does matter: 1.5 Fr. stone baskets almost double irrigation flow during flexible ureteroscopy compared to 1.9 Fr. Stone baskets Clinical Summary Send a SMS Share
Ordering information
For ordering information, please call 800-258-3476.