Altis® 522 Post-Market Surveillance Study
Prospective, post-market, multicenter, comparative cohort study
The first and only 522 study to include a single incision sling and full-length retropubic and transobturator slings.
The Altis 522 study, which compares Altis to FDA-cleared retropubic and transobturator slings, was designed to reflect real-world clinical practice.
Quick Links: Product Information | Primary Effectiveness | Secondary Effectiveness | Subjective Outcomes
Safety & Efficacy | Important Safety Information
The single incision sling system that delivers for you and your patients.
The Altis® Single Incision Sling System is a unique, minimally invasive solution purposefully designed to provide predictable placement and adjustable control. This makes the procedure straightforward, accurate and repeatable.1
- Patented helical introducer makes the surgical procedure straightforward, accurate and reproducible.2
- Dynamic anchor allows for precise tensioning.2
- Patented, lightweight mesh features low elasticity for stable support.2


Purposefully designed for a better patient experience.
The Altis® system allows for a more convenient procedure that can be performed in an outpatient setting under local anesthesia, which may lead to a faster recovery time. Patients can be back on their feet and return to work or normal activity sooner as compared to traditional full-length slings.1,2,3
At 24-Months, the Altis® Single Incision Sling is Comparable to Full-Length Retropubic and Transobturator Slings
24-Month results are available for review.
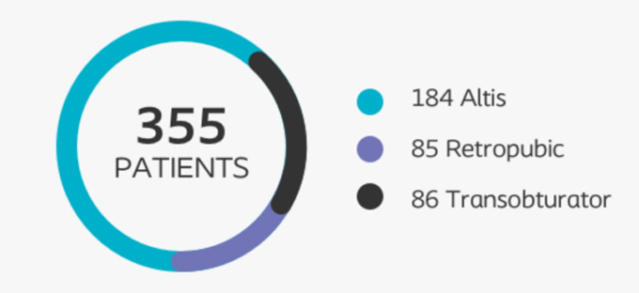
The Altis® 522 study is a prospective, post-market, multicenter, comparative cohort study performed in adult female patients with SUI who are clinically indicated for mid-urethral sling surgery.
Consistent Results
At 24 months, 24-hour pad weight success (≥50% reduction), negative CST, PGI-I, UDI-6, and IIQ-7 were similar between Altis and full-length retropubic and transobturator slings.
Altis product design (i.e., elastic properties similar to the pubourethral ligament and intraoperative adjustability post-deployment) and surgical reproducibility attributed to high objective and subjective cure rates at 24 months, comparable to full-length retropubic and transobturator slings.
Rigorous Testing
The Altis 522 study was designed to have a full range of objective and subjective outcome measurements, included full-length retropubic and transobturator slings in the comparator arm, and did not exclude ISD patients.
The same rigorous and robust protocol and testing methods utilized in Altis IDE study were included in the Altis 522 study.
Gold Standard Comparisons
All commercially available full-length retropubic and transobturator slings were allowed in the comparator group.
By allowing a heterogenous group of full-length retropubic and transobturator slings in the comparator arm, and enrolling patients with a variety of characteristics, the Altis 522 study is more reflective of real world clinical practices.
Ongoing Commitment
The aim of the Altis 522 study is to compare the safety and effectiveness of the Altis Single Incision Sling to full-length retropubic and transobturator slings through 36 months.
Subsequent reports will follow at 36 months.
Primary Effectiveness Objective
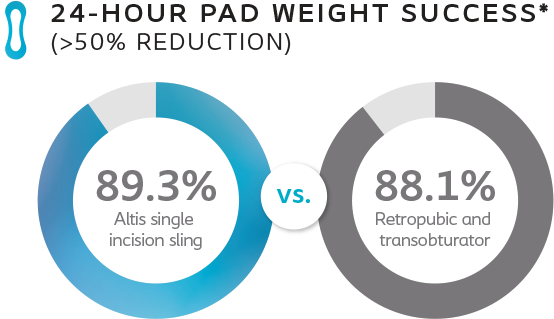
*Results compared to baseline collected at 24 months.
The primary efficacy objective was to demonstrate that the reduction from baseline in 24-hour pad weight associated with Altis SIS is non inferior to the rate associated with the use of transobturator and/or retropubic slings at 6 months.
At 24 months, Altis is comparable to full-length retropubic and transobturator slings.
Secondary Effectiveness Objective
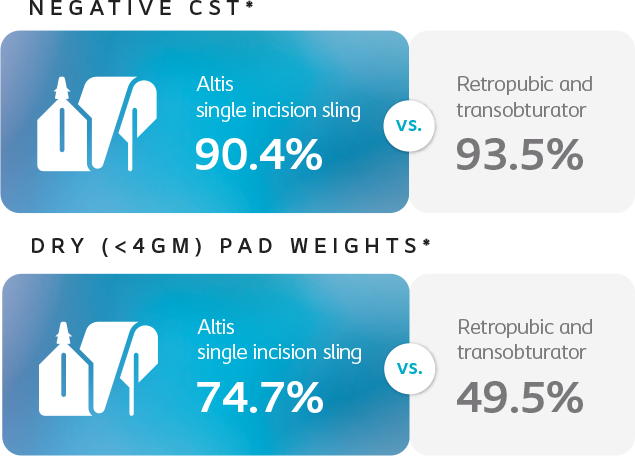
*Results compared to baseline collected at 24 months.
Secondary efficacy measures included objective dryness(defined as pad weight ≤4.0g) and negative CST with comparative assessments between groups at 6, 12, 18, 24 and 36 months.
At 24 months, Altis is comparable to full-length retropubic and transobturator slings.
Subjective Outcomes
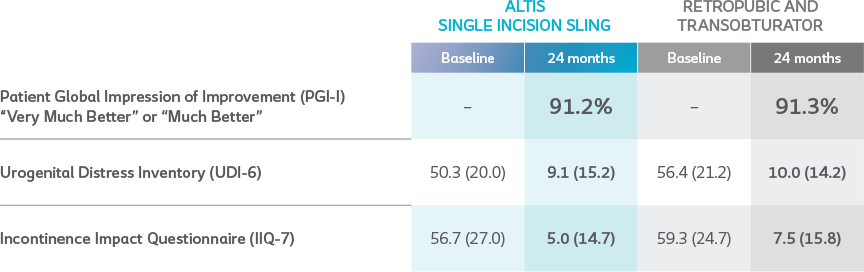
*Results compared to baseline collected at 24 months.
Subjective outcome measures collected included patient global impression of improvement (PGI-I), urogenital distress inventory (UDI-6), Incontinence Impact Questionnaire − Short Form (IIQ-7), Surgical Satisfaction Questionnaire (SSQ-8), and visual analog scale for pain (VAS). At 24 months, Altis is comparable to full-length retropubic and transobturator slings.
Safety and Efficacy Outcome Measures
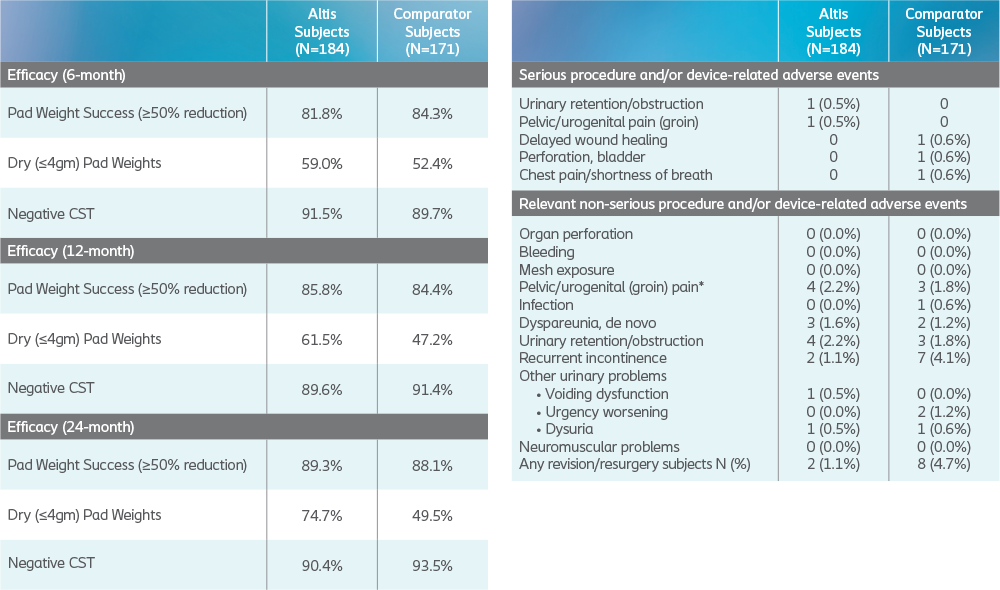
No new serious procedure – and/or device-related adverse events were reported in either study arm since the previous 12-month publication.
